Brain metastases
Brain metastases represent more than half of intracranial tumors. They are diagnosed in 10% to 30% of adults and 6% to 10% of children with malignant tumors. 98.000 to 170.000 new cases are recorded each year (Johnson et al. 1996) in the United States. In approximately 20% of patients, the brain lesions are the only manifestation of metastatic tumor, while at 80% extracerebral metastases were diagnosed.
Lung cancer, malignant melanoma, breast cancer, renal cell cancer and gastro intestinal cancer are the most common tumor entities, which lead to a brain metastasis.

Morphology
About two-thirds of intracranial metastases are supratentorial. Primary tumors of the pelvis and the preferred gastrointestinal tract tend to infratentorial metastasis. The reasons for this phenomenon are unclear. Brain metastases are rounded or oval configured lesions displace the healthy tissue and do not grow infiltrative usually. They are preferably present in the boundary region between the white and gray matter in the field of vascular boundary lines (Russel and Rubinstein, 1989;. Hwang et al 1996).

Frequency
Brain metastases represent more than half of intracranial tumors. They are diagnosed in 10 to 30% of adults and 6 to 10% of children with malignant tumors. Between 98,000 and 170,000 new cases are registered in the USA each year (Johnson et al. 1996). In about 20% of the patients, the brain lesions represent the only metastatic tumor manifestation, while extracerebral metastases were also diagnosed in 80%.
Bronchial carcinoma, malignant melanoma, breast carcinoma, hypernephromas and colorectal carcinoma are the most common tumor entities leading to brain metastasis. Head and neck tumors, andrological and gynecological tumors as well as bone and soft tissue sarcomas rarely metastasize intracranially (Mehta et al. 1992).

Clinical symptoms
Two-thirds of patients with brain metastases develop neurological problems: headaches, seizures, focal neurological deficits, signs of intracranial pressure increase, signs of frontal lobe syndrome, cognitive disorders, etc. The symptoms may occur slowly progressive or acute, as in stroke, especially if a vascular events metastasis accompanied:
Bleeding (in particular melanoma, renal carcinomas or thyroid carcinomas) or ischemia, by tumor compression or embolization. These symptoms must be differentiated from neurological deficits of other causes, such as side effects of therapy (chemotherapy, radiotherapy or surgery), paraneoplastic syndromes, disorders caused by spinal compression, carcinomatous meningitis etc.

Diagnostics
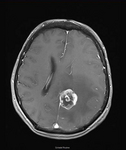
Brain metastases are characterized by a pathological blood-brain barrier and documented in imaging (CT and MRI) as a strong contrast enhancing structures with perifocal edema. The MRI shows a higher sensitivity than CT, especially the T1 weighted images for increased gadolinium application. As a differential diagnosis brain intrinsically tumors, inflammatory diseases or meningiomas can be discussed. Functional studies such as PET, SPECT and MR spectroscopy may be useful for the differential diagnosis of tumor versus necrosis after radiotherapy. A complete current staging investigation is very important for the choice of one or the other therapeutic strategy.
The biopsy is indicated in patients with unclear radiological findings, with unknown primary tumor or with unclear oncologic history, eg at a large time interval between the diagnosis of the primary tumor and brain metastasis.

Therapy
Microsurgery, radiosurgery (Cyberknife), whole brain radiotherapy (WBRT), stereotactic fractionated radiotherapy (FSRT), chemotherapy and sole symptomatic treatment are the main therapeutic options. The final decision depends on the clinical neurological status of the patient, the activity of the primary tumor and the number and localization of metastases.
Symptomatic therapy
The sole symptomatic therapy with cortisone reduces the perifocal edema and alleviates the neurological symptoms temporarily. The preparations with dexamethasone, as compared to the other corticosteroids, the advantage of a lower risk of adverse reactions (edema, hypokalemia, infections, psychosis, etc.). The dosage depends on the clinical symptoms and extent of the edema. In patients with seizures an antiepileptic drug is indicated.
Radiosurgery (Cyberknife, ZAP-X)
Brain metastases with a maximum diameter less than 3 cm are an ideal target for the CyberKnife or ZAP therapy. The local tumor control is about 90%. Scientific data have shown that in the treatment of selected diverse patients with brain metastases, radiosurgery is at least equivalent to a much greater patient comfort (Muacevic 2005).
In general, it was observed that the histology of the primary tumor for tumor control and response after radiosurgery has no statistical significance (Grosu et al. 2000). So-called radiation-resistant entities such as malignant melanoma and hypernephroma show a very good local tumor control after single irradiation (Engenhart et al 1993;. Alexander et al., 1995). The main side effect of the therapy, the radiation reaction, occurring at a frequency of about 2% -5% occurs and is usually treated with cortisone.
Microsurgery
There is a clear indication for surgical therapy in the case of superficial, larger metastases or tumors that cause progressive neurological symptoms due to mass displacement. In foci in the posterior fossa, the size of the metastasis or a pronounced perifocal edema may pose a risk of cerebral herniation or hydrocephalus occlusus. Emergency surgical removal of the metastasis is indicated here (Reulen et al. 1995).
Regardless of the type of therapy being chosen for the overall prognosis, in addition to the control of the primary tumor, the clinical condition of the patient before treatment is critical. Patients in a good clinical condition survive in application of both possible methods (surgery or radiotherapy) was significantly longer than patients in worse clinical status (Karnofsky performance score <70). Other prognostic factors such as patient age, tumor location, type of primary tumor and the latency period between diagnosis of the primary tumors and occurrence of cerebral metastasis play a minor role (Muacevic et al. 1999).
Metastases of small cell lung cancer, malignant lymphoma and germ cell tumors should be treated neither surgically nor radiosurgery, unless they cause a clinically significant compression. In the treatment are generally GSB and chemotherapeutic method in the foreground.
A retrospective analysis of Bindal et al. (1993) showed that patients with multiple metastases can be successfully treated by surgery and can achieve survival rates to those of patients with singular metastases. A more invasive procedure should not be recommended except for rare cases with very good clinical condition and convenient lesions.
Whole brain irradiation
WBRT is the method of choice in many cerebral metastasis. The role of WBRT as adjunctive therapy after or before radiosurgery is controversially discussed in the literature. The possibility of a progression of brain metastasis through regular MRI studies (in the first 3 years, every 3-4 months) to discover in time and the overall prognosis of the patient should play an important role in the decision-making process.
After WBRT a complete alopecia may occur, which is usually reversible. In isolated cases, long-term consequences like cognitive impairment to dementia (DeAngelis et al. 1989) is described. Morphological changes such as atrophy or leukoencephalopathy are observed on the MRI scans often after therapy, but remained usually without clinical correlation.
Chemotherapy
Chemotherapy has been in the treatment of patients with BM yet no clearly defined role in the treatment plan. The assumption that the blood-brain barrier prevents the penetration of chemotherapeutic agents in the affected areas in the CNS, was the reason to use the radiation in the presence of brain metastases and preferred to give chemotherapy rarely or never preference. However, it has been shown in some clinical studies that the blood-brain barrier is not the decisive factor that thwarted a successful chemotherapy of brain metastases. Chemotherapy regimens have been used successfully for brain metastases of systemically untreated patients with small cell lung cancer (Kristensen et al., 1992), breast cancer (Boogerd et al. 1992) and germ cell tumors (Fossa et al. 1999). When using the usual chemotherapy regimens tumor regression rates were the same as not inferior to exclusively extracranial metastasis and radiotherapy. In a review article the results of 12 patient series of 116 patients with metastatic small cell lung cancer combined (Kristensen et al. 1992).


